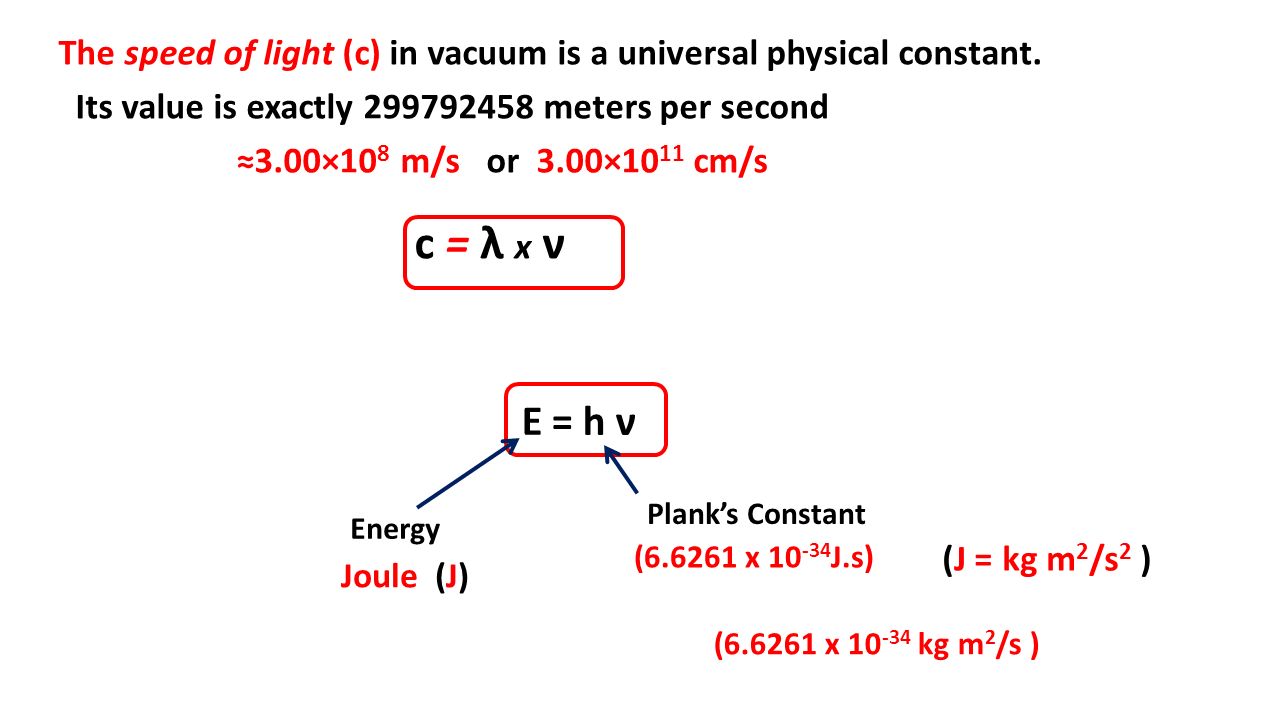
The values of Planck's constant is 6.63 × 10^-34 Js. The velocity of light is 3.0 × 10^8 ms^-1 . Which value is closest to the wavelength in nanometres of a quantum of light which frequency of 8 × 10^15s^-1 ?

Verify that the correct value for the speed of light c is obtained when numerical values for the permeability and permittivity of free space ( µ0 and ε0 ) are entered into
Would light travel faster in a vacuum with no quantum fluctuations? Would this be the real speed limit of the universe that not even tachyons can outspeed? - Quora

The value of Planck's constant is 6.63 x 10-34 Js. The speed of light is 3x10*7 nm 5-1. Which value is closest to the wavelength in nanometer of a quantum of light

The values of Planck's constant is 6.63 × 10^-34 Js. The velocity of light is 3.0 × 10^8 ms^-1 . Which value is closest to the wavelength in nanometres of a quantum of light which frequency of 8 × 10^15s^-1 ?

The value of Planck's constant is 6.63 x10-34 Js. The velocity of light is 3.0 x108 ms -1. Which value is closest to the wavelength in nanometers of a quantum of light
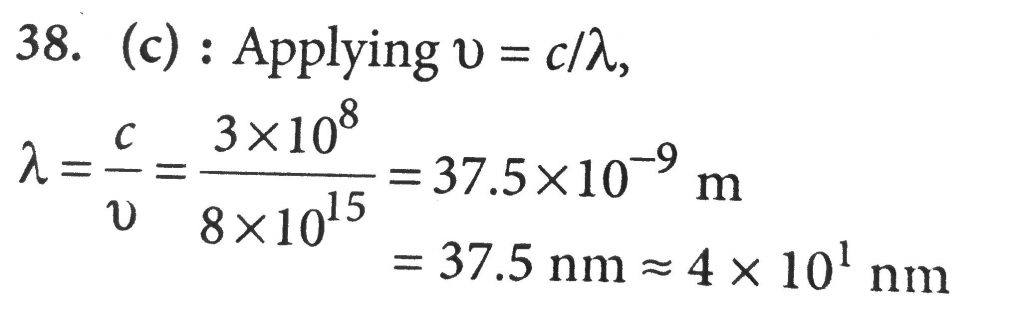
The value of Planck's constant is 6.63 x 10^-34 Js. The velocity of light is 3.0 x 10^8 ms^-1. Which value is closest to the wavelength in metres of a quantum of

The velocity of light in diamond is 1,21,000 km s-1. The refractive index of the diamond is (Velocity of Light in air = 3 x 108 m s-1).

The value of Planck's constant is 6.63 x 104JS. The speed of light is 3x107 nm s 1. Which value is closest to the wavelength in nanometer of a quantum of light