![SOLVED:Consider the multistep reaction. What is the best rate aw for the overall reaction? A) Rate k[AJ[BJIC] B) Rate kz[C] 2A C D (slow) Rate k[AJ?[B] 2E (fast) D) Rate (ki[AJ[BI)(kz[C]) E) SOLVED:Consider the multistep reaction. What is the best rate aw for the overall reaction? A) Rate k[AJ[BJIC] B) Rate kz[C] 2A C D (slow) Rate k[AJ?[B] 2E (fast) D) Rate (ki[AJ[BI)(kz[C]) E)](https://cdn.numerade.com/ask_images/2522bd6105e148b496e4b3fed701912d.jpg)
SOLVED:Consider the multistep reaction. What is the best rate aw for the overall reaction? A) Rate k[AJ[BJIC] B) Rate kz[C] 2A C D (slow) Rate k[AJ?[B] 2E (fast) D) Rate (ki[AJ[BI)(kz[C]) E)

The Rate Law. Objectives: To understand what a rate law is To determine the overall reaction order from a rate law CLE ppt download
![OneClass: The rate of a certain reaction is given by the following rate law: 2 rate-k [N2]2[ rate-k N... OneClass: The rate of a certain reaction is given by the following rate law: 2 rate-k [N2]2[ rate-k N...](https://prealliance-textbook-qa.oneclass.com/qa_images/homework_help/question/qa_images/130/13037717.png)
OneClass: The rate of a certain reaction is given by the following rate law: 2 rate-k [N2]2[ rate-k N...
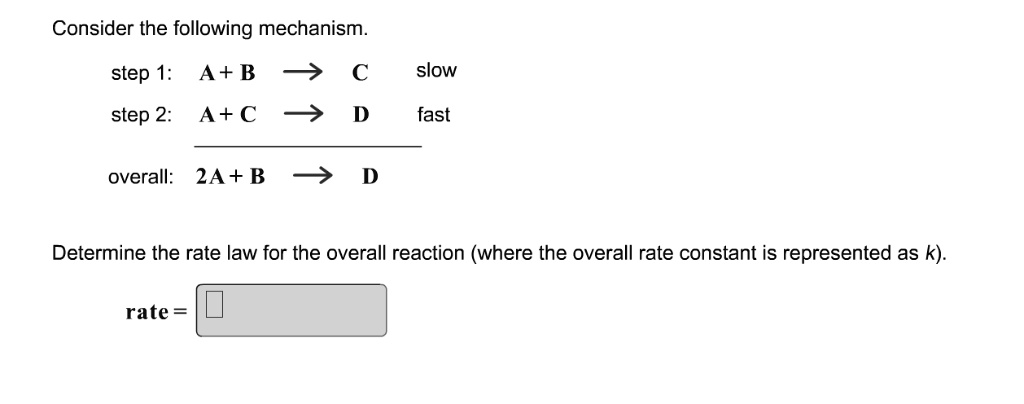
SOLVED:Consider the following mechanism_ step 1: A+ B slow step 2: A+C D fast overall: 2A + B D Determine the rate law for the overall reaction (where the overall rate constant
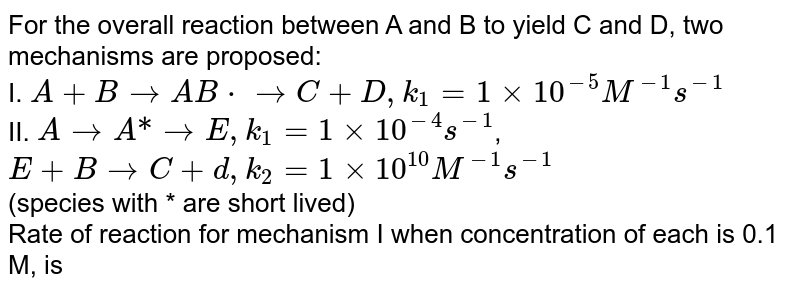
For the overall reaction between A and B to yield C and D, two mechanisms are proposed: I. A+BtoAB*toC+D,k_(1)=1xx10^(-5)M^(-1)s^(-1) II. AtoA"*"toE,k_(1)=1xx10^(-4)s^(-1), E+BtoC+d,k_(2)=1xx10^(10)M^(-1)s^(-1) (species with * are short lived) AT what ...



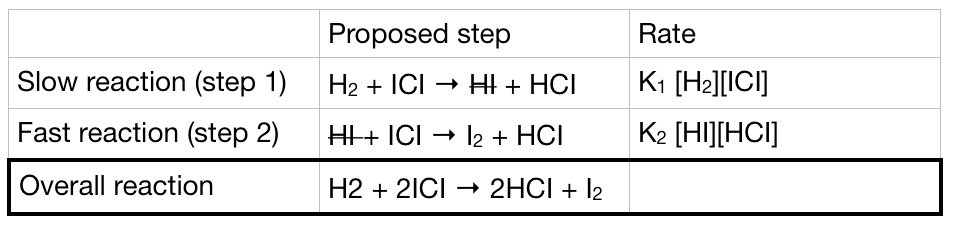







![For Rate = k[A]2[B]3, determine the order ... | Clutch Prep For Rate = k[A]2[B]3, determine the order ... | Clutch Prep](https://lightcat-files.s3.amazonaws.com/problem_images/2a47d576544d4e1c-1539199742539.jpg)


